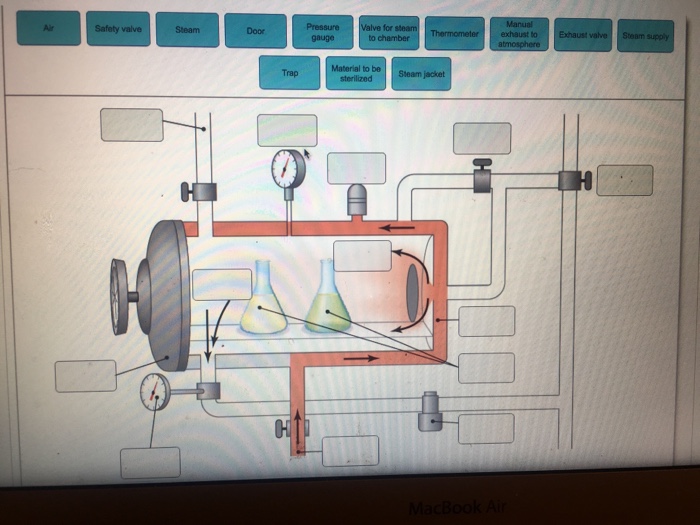
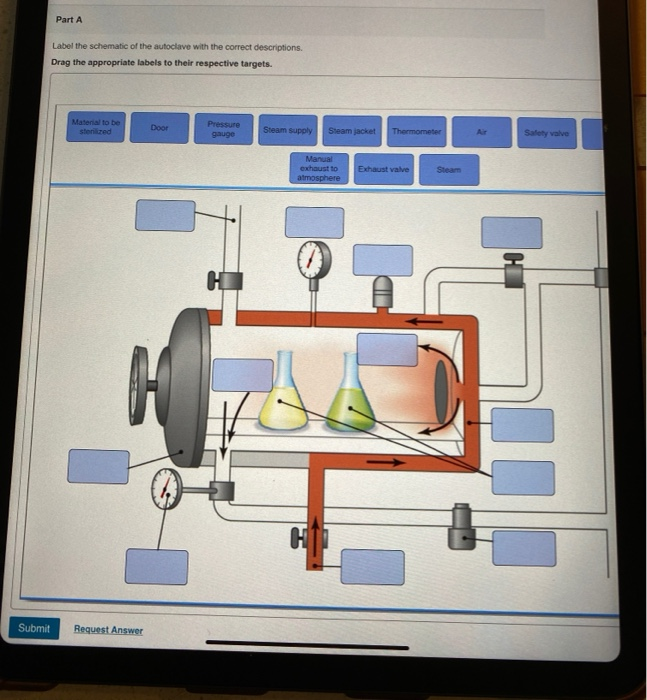
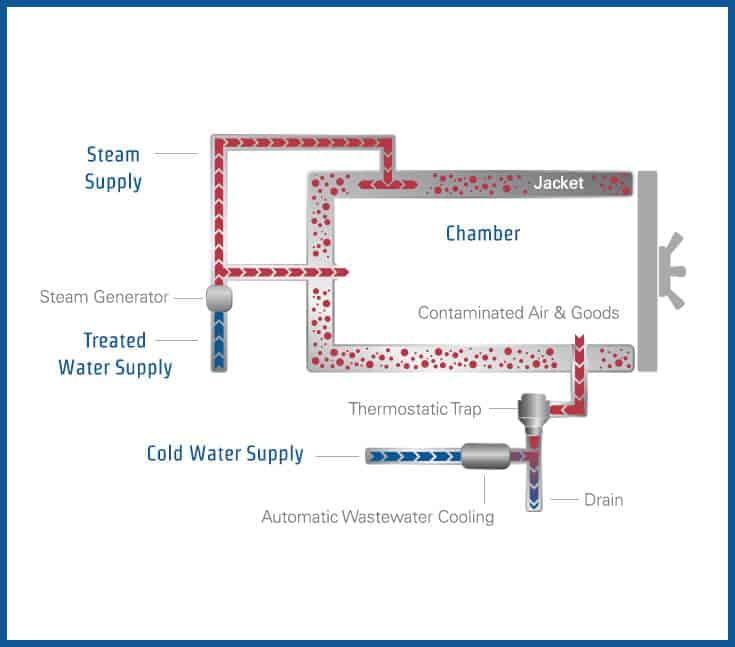
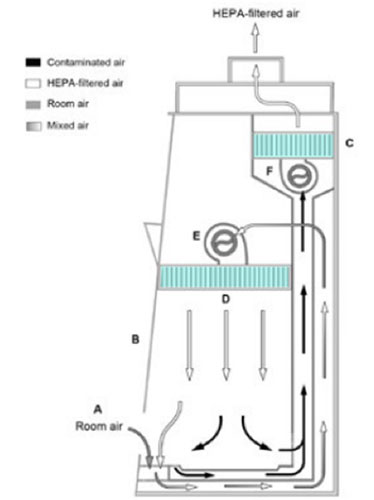
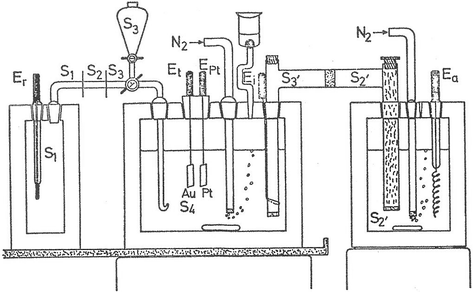
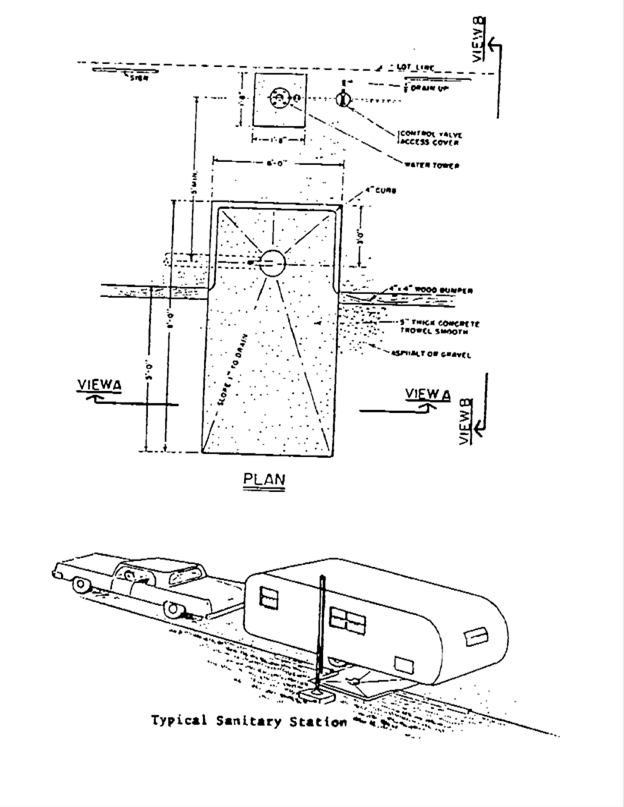
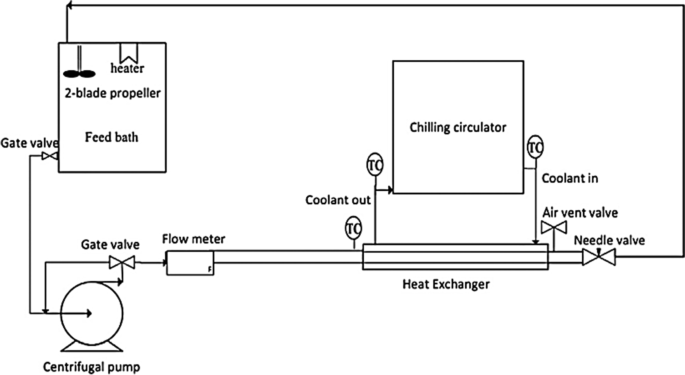
40 label the schematic of the autoclave with the correct descriptions
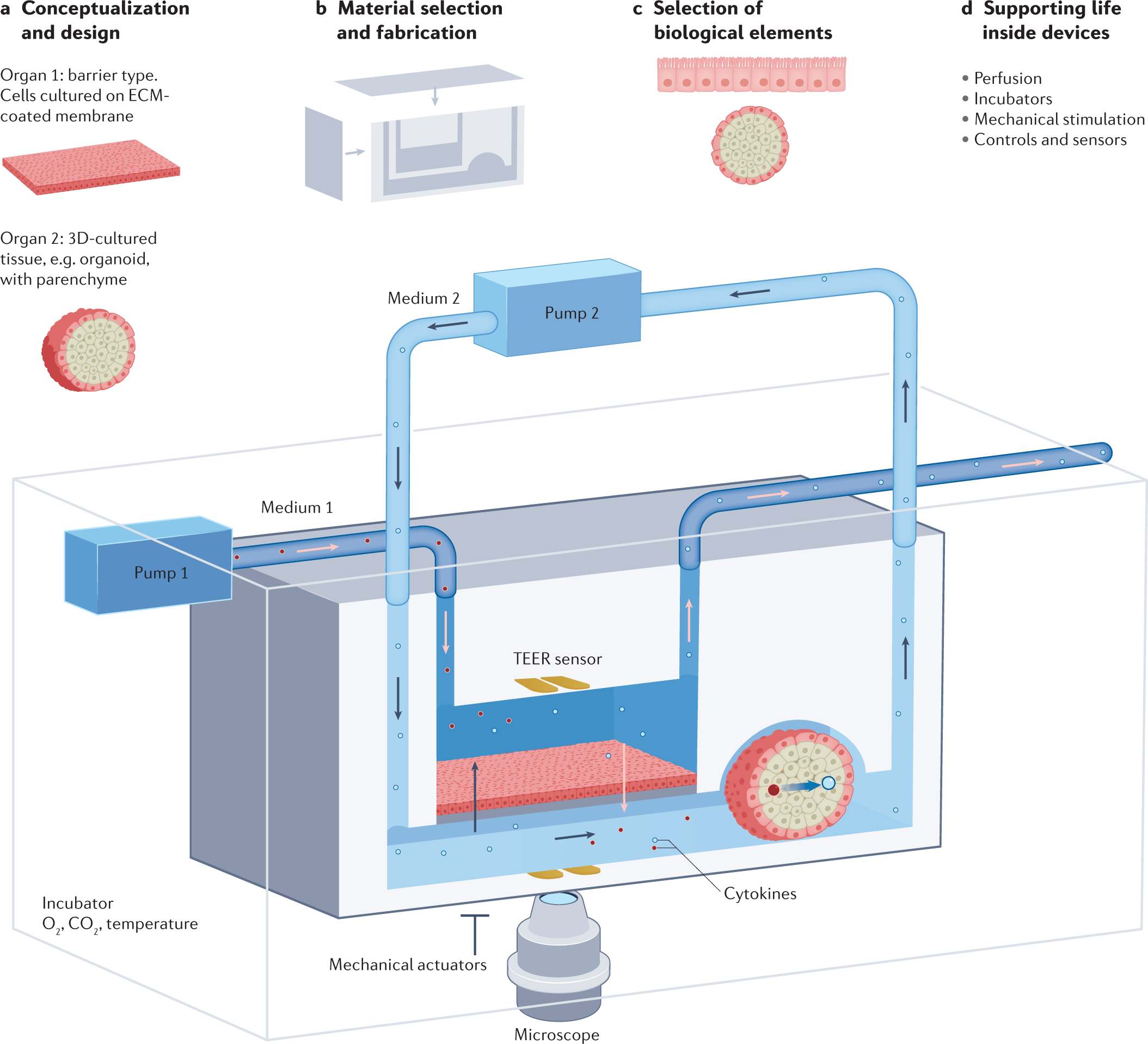
Molecular Cloning: A Laboratory Manual Third Edition 23.8.2000 · The emergence of recombinant DNA technology occurred via the appropriation of known tools and procedures in novel ways that had broad applications for analyzing and modifying gene structure and organization of complex genomes. Handbook for Sampling and Sample Preservation of Water and … ----- FOREWORD Environmental measurements are required to determine the quality of ambient waters and the character of waste effluents. The Environmental Monitoring and Support Laboratory - Cincinnati: Develops and evaluates techniques to measure the pres.ence and centration of physical, chemical, and radiological pollutants con- water, wastewater, bottom sediments, and solid waste.
Documentation and Records: Harmonized GMP Requirements - PMC The batch production record should be checked before issuance to assure that it is the correct version and a legible accurate reproduction of the appropriate master production instruction. If the batch production record is produced from a separate part of the master document, that document should include a reference to the current master production instruction being used.

Label the schematic of the autoclave with the correct descriptions
Bioprocess Engineering Principles-Pauline M. Doran Chemical Engineering Research and Design. Bioreactor Retrofitting to Avoid Aeration with Oxygen in Pichia Pastoris Cultivation Processes for Recombinant Protein Production (PDF) Bioprocess Engineering-Basic Concepts by Shuler and Kargi ... Bioprocess Engineering-Basic Concepts by Shuler and Kargi ( PDFDrive ) Education Development Center data:image/png;base64,iVBORw0KGgoAAAANSUhEUgAAAKAAAAB4CAYAAAB1ovlvAAAAAXNSR0IArs4c6QAAArNJREFUeF7t1zFqKlEAhtEbTe8CXJO1YBFtXEd2lE24G+1FBZmH6VIkxSv8QM5UFgM ...
Label the schematic of the autoclave with the correct descriptions. Manual of Patent Practice - Section 14: The application - GOV.UK If the applicant wishes to correct his or her name, then Form 20 is required. However, correction of any of the other information given on Form 1 merely requires notification in writing. ( See 19. ... Chemical Engineering Design Principles Practice and Chemical Engineering Design Principles Practice and Economics of-Plant and Process Design GMP Standard Operating Procedures (SOPs) This process has schematic blueprints for your knowing of how different types of records are prepared and saved in a common data source. Quality Documentation Change Control. This procedure describes the role of manufacturing personnel in creating and reviewing/updating Quality Documentation, Document Control Officers, File Administrators. Comprehensive protocols for CRISPR/Cas9-based gene editing in … Correct targeting is confirmed by PCR screening. An additional subcloning step in which a recombinase is transiently introduced is required ... Using a label maker (not by ... drug screening, and cell transplantation. In the context of hPSCs, one of the first descriptions of CRISPR/Cas9-based gene editing was at the “safe harbor” AAVS1 ...
Handbook of Modern Pharmaceutical Analysis - Academia.edu Now a day the control of pharmaceutical impurities in industry is a critical issue and these impurities can be API related impurities, process related impurities or stability related impurities, hence ICH has formulated guidelines regarding the control of impurities. Education Development Center data:image/png;base64,iVBORw0KGgoAAAANSUhEUgAAAKAAAAB4CAYAAAB1ovlvAAAAAXNSR0IArs4c6QAAArNJREFUeF7t1zFqKlEAhtEbTe8CXJO1YBFtXEd2lE24G+1FBZmH6VIkxSv8QM5UFgM ... (PDF) Bioprocess Engineering-Basic Concepts by Shuler and Kargi ... Bioprocess Engineering-Basic Concepts by Shuler and Kargi ( PDFDrive ) Bioprocess Engineering Principles-Pauline M. Doran Chemical Engineering Research and Design. Bioreactor Retrofitting to Avoid Aeration with Oxygen in Pichia Pastoris Cultivation Processes for Recombinant Protein Production
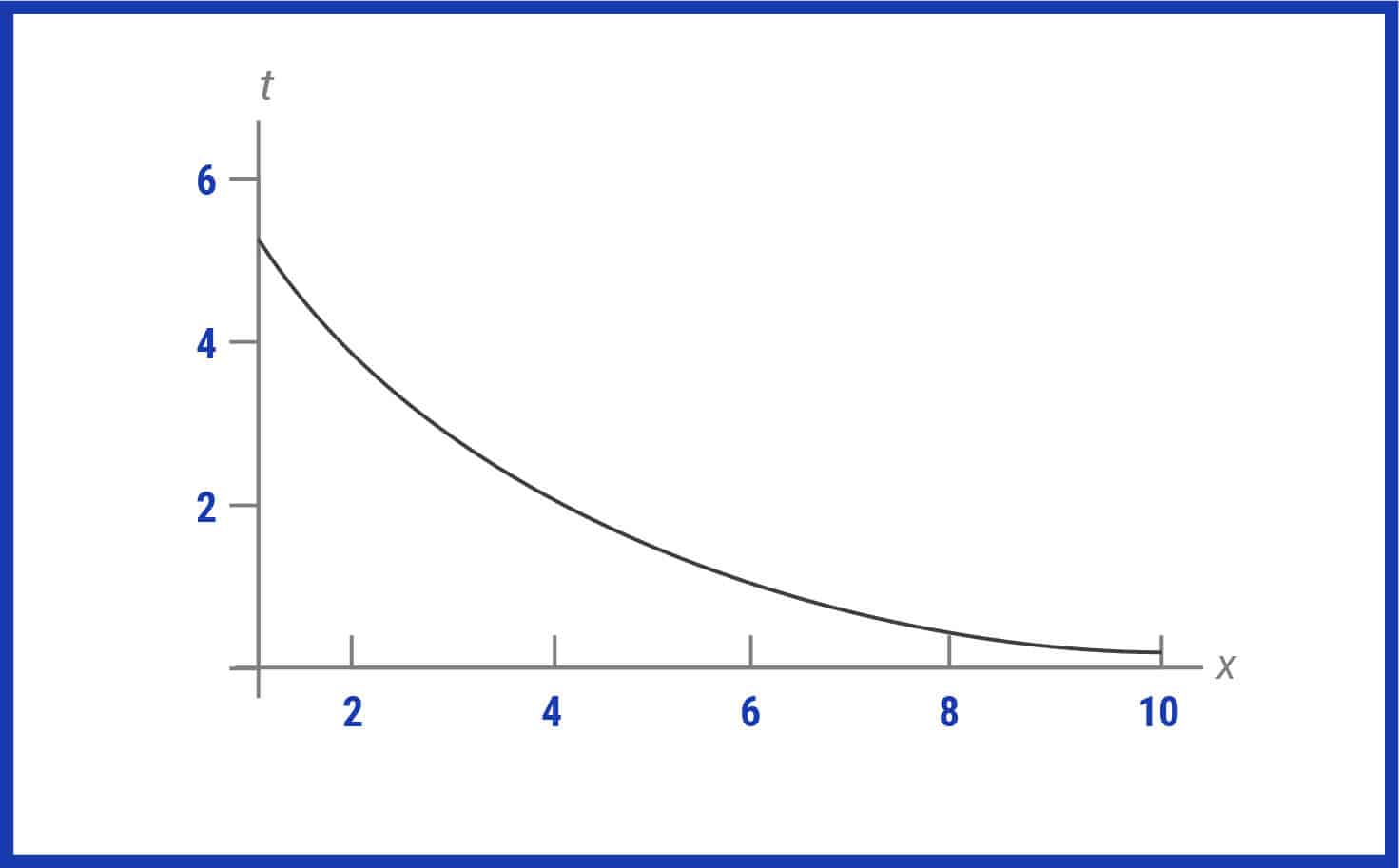
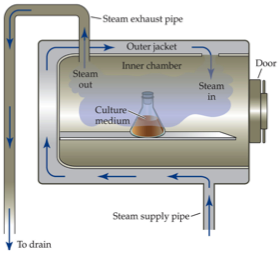

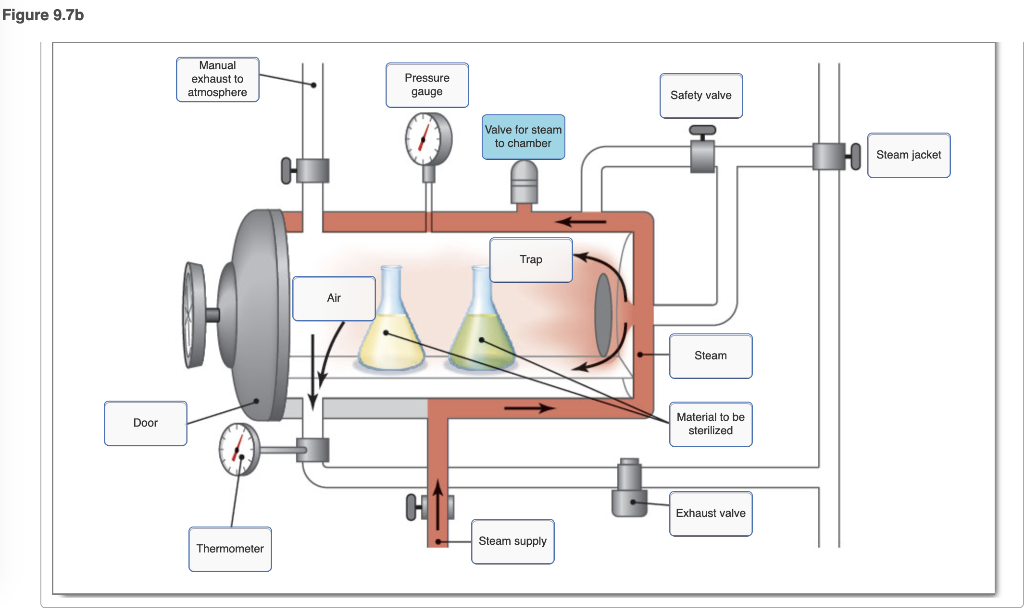
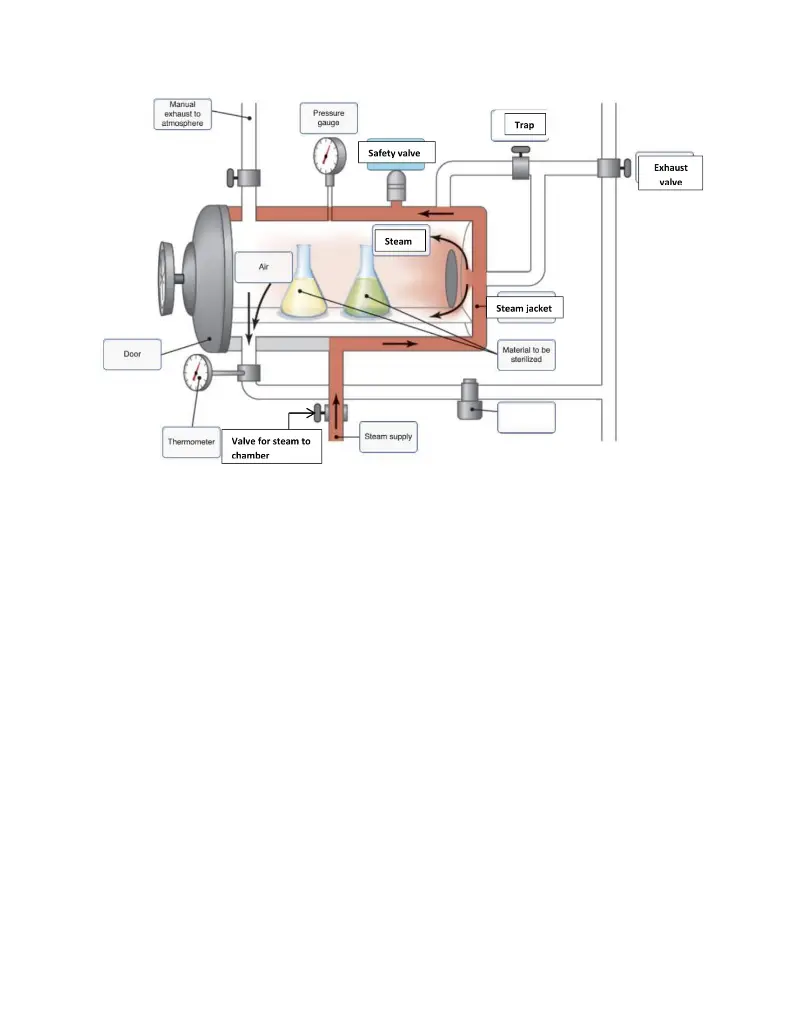














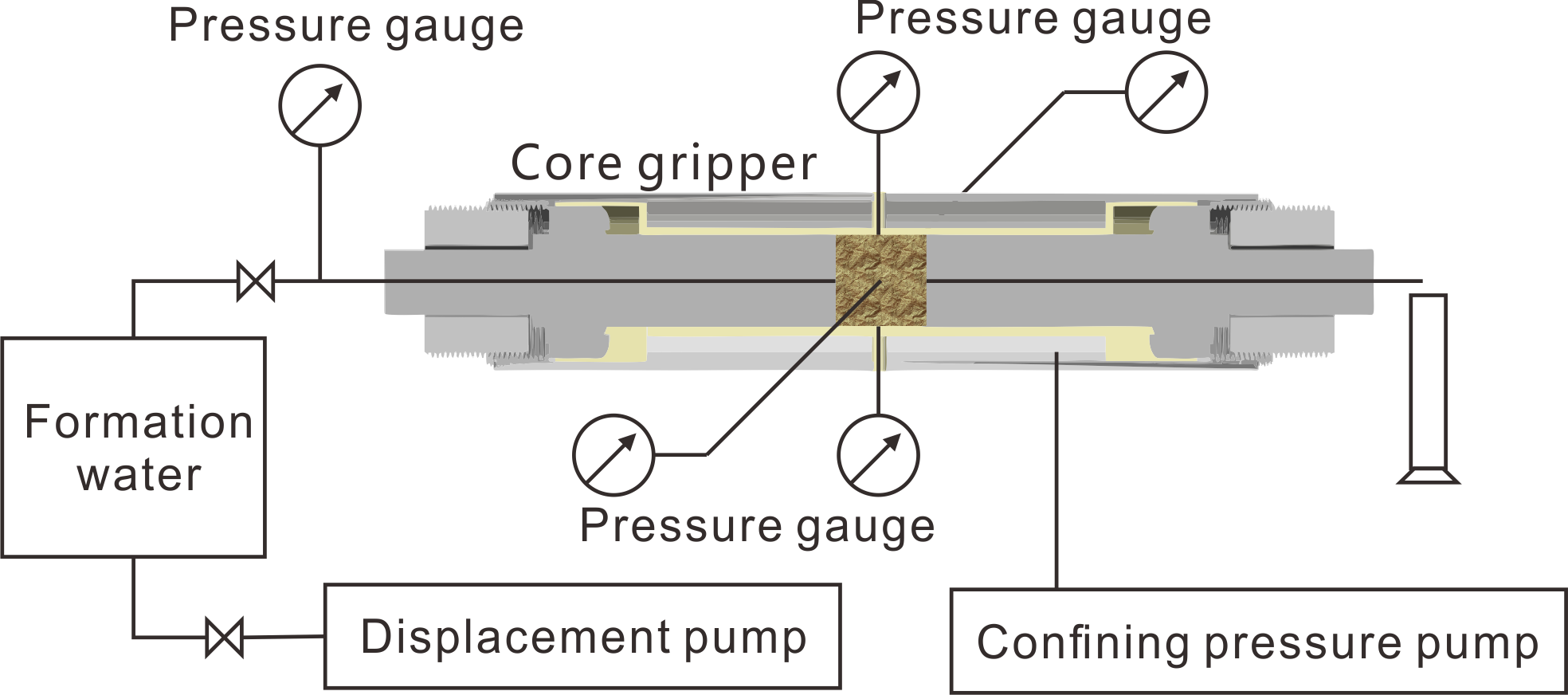
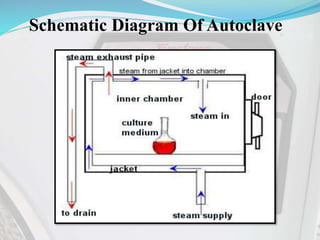
Post a Comment for "40 label the schematic of the autoclave with the correct descriptions"